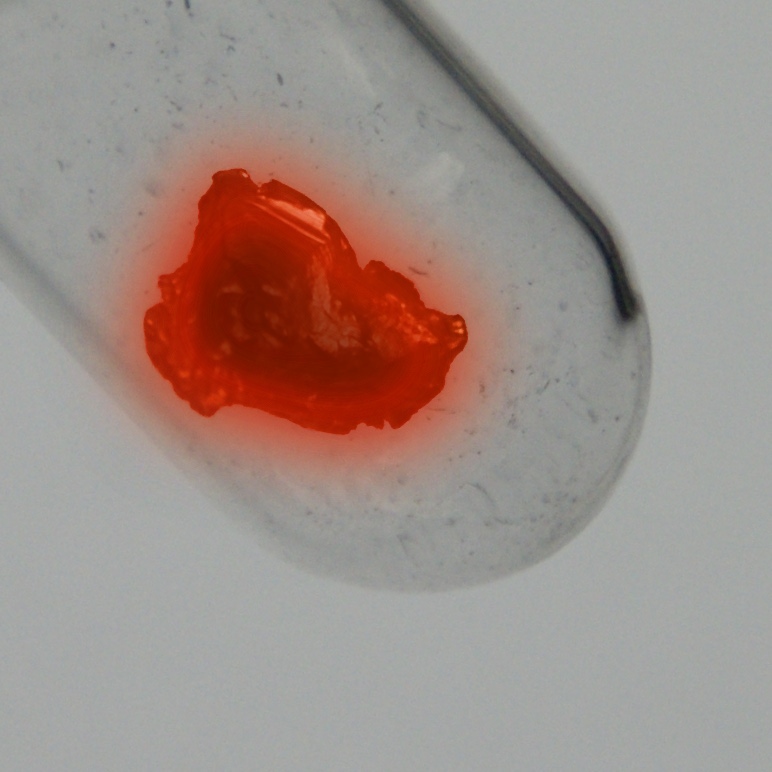
Protactinium
91
Pa
Group
n/a
Period
7
Block
f
Protons
Electrons
Neutrons
91
91
140
General Properties
Atomic Number
91
Atomic Weight
231.03588
Mass Number
231
Category
Actinides
Color
Silver
Radioactive
Yes
From the Greek protos meaning first
Crystal Structure
Centered Tetragonal
History
In 1900, William Crookes isolated protactinium as an intensely radioactive material from uranium
Protactinium was first identified in 1913 by Kasimir Fajans and Oswald Helmuth Göhring in Germany.
A more stable isotope of protactinium was discovered in 1917 by Otto Hahn and Lise Meitner at the Kaiser Wilhelm Institute in Berlin.
Protactinium was first identified in 1913 by Kasimir Fajans and Oswald Helmuth Göhring in Germany.
A more stable isotope of protactinium was discovered in 1917 by Otto Hahn and Lise Meitner at the Kaiser Wilhelm Institute in Berlin.
Electrons per shell
2, 8, 18, 32, 20, 9, 2
Electron Configuration
[Rn] 5f2 6d1 7s2
Protactinium is one of the rarest and most expensive naturally occurring elements
Physical Properties
Phase
Solid
Density
15.37 g/cm3
Melting Point
1841.15 K | 1568 °C | 2854.4 °F
Boiling Point
4300.15 K | 4027 °C | 7280.6 °F
Heat of Fusion
15 kJ/mol
Heat of Vaporization
470 kJ/mol
Specific Heat Capacity
-
Abundance in Earth's crust
9.9×10-13%
Abundance in Universe
n/a
CAS Number
7440-13-3
PubChem CID Number
n/a
Atomic Properties
Atomic Radius
163 pm
Covalent Radius
200 pm
Electronegativity
1.5 (Pauling scale)
Ionization Potential
5.89 eV
Atomic Volume
15.0 cm3/mol
Thermal Conductivity
0.47 W/cm·K
Oxidation States
3, 4, 5
Applications
Owing to its scarcity, high radioactivity and high toxicity, there are currently no uses for protactinium outside of scientific research.
With the advent of highly sensitive mass spectrometers, an application of 231Pa as a tracer in geology and paleoceanography has become possible.
Protactinium-231 combined with the thorium-230 can be used to date marine sediments.
With the advent of highly sensitive mass spectrometers, an application of 231Pa as a tracer in geology and paleoceanography has become possible.
Protactinium-231 combined with the thorium-230 can be used to date marine sediments.
Protactinium is toxic and highly radioactive
Isotopes
Stable Isotopes
-Unstable Isotopes
212Pa, 213Pa, 214Pa, 215Pa, 216Pa, 217Pa, 218Pa, 219Pa, 220Pa, 221Pa, 222Pa, 223Pa, 224Pa, 225Pa, 226Pa, 227Pa, 228Pa, 229Pa, 230Pa, 231Pa, 232Pa, 233Pa, 234Pa, 235Pa, 236Pa, 237Pa, 238Pa, 239Pa, 240Pa